Revolutionary breakthrough: seawater turns into hydrogen directly! Australian magic catalyst produces hydrogen for only 2 Australian dollars per kilogram
Seawater turns into hydrogen directly! Yes, you read that right. Recently, researchers from the Royal Melbourne Institute of Technology in Australia developed a magic catalyst that can produce hydrogen directly in seawater, with an estimated cost of only 2 Australian dollars (about 10 yuan) per kilogram, much lower than the current cost of hydrogen production from fossil fuels.
Hydrogen is a clean energy carrier that can be used in multiple sectors such as transportation, industry, construction, power and more. It can even be used for boiling water, cooking, heating and so on. It helps us achieve a low-carbon economy and cope with climate change.
However, most of the hydrogen in the world today is made by converting fossil fuels, emitting 830 million tons of carbon dioxide every year, causing huge environmental pollution.
The current “green” hydrogen is not green either. It requires electrolyzing freshwater to produce, which is very expensive and almost commercially unfeasible. It only accounts for 1% of the global total hydrogen production.
Therefore, finding a sustainable, low-cost and efficient way to produce hydrogen is a matter of urgency.
The ocean covers 71% of the Earth’s surface, and scientists have always hoped to use seawater to produce hydrogen. However, seawater contains a lot of salt and impurities, such as sodium, magnesium, calcium, potassium, chlorine, sulfate and so on. These substances interfere with the process of seawater electrolysis for hydrogen production, causing problems such as catalyst deactivation, electrode corrosion, membrane clogging and even toxic chlorine gas release.
Therefore, the biggest pain point of seawater hydrogen production is to find a special catalyst that can effectively catalyze the water splitting reaction, resist the corrosion and pollution in seawater, and be absolutely cheap to reduce costs.
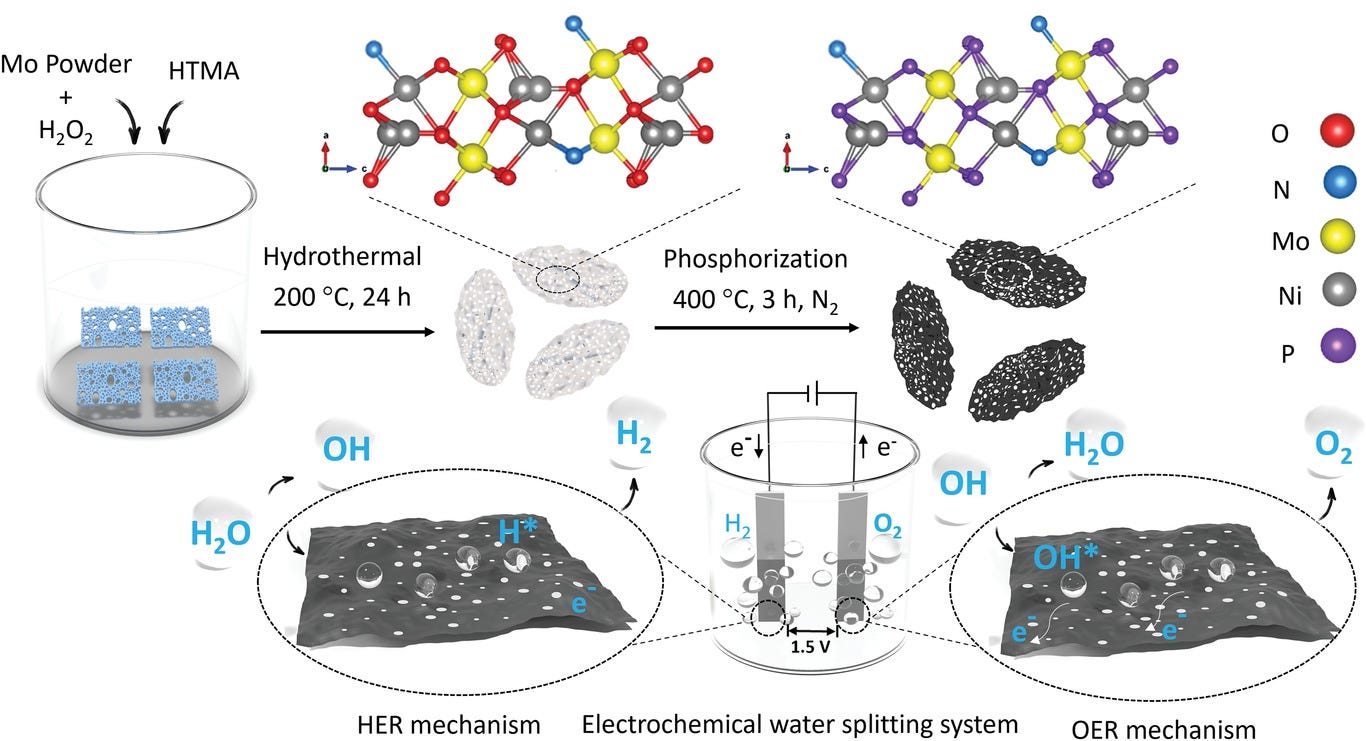
The researchers from the Royal Melbourne Institute of Technology developed such a magic catalyst: porous N-NiMo3P nanosheets.
These sheets have large uniform pores on the base surface, which have higher catalytic activity and ensure faster mass transfer. They can effectively catalyze the seawater electrolysis reaction at room temperature, directly splitting seawater into hydrogen and oxygen without desalination.
The most critical thing is that using these sheets, the overpotentials and total voltage of both hydrogen evolution reaction and oxygen evolution reaction are very low, which means that the catalytic efficiency is very high and the electric energy consumption is very low.
The most most critical thing is that this catalyst can catalyze complete water splitting, that is, completely split water into hydrogen and oxygen.
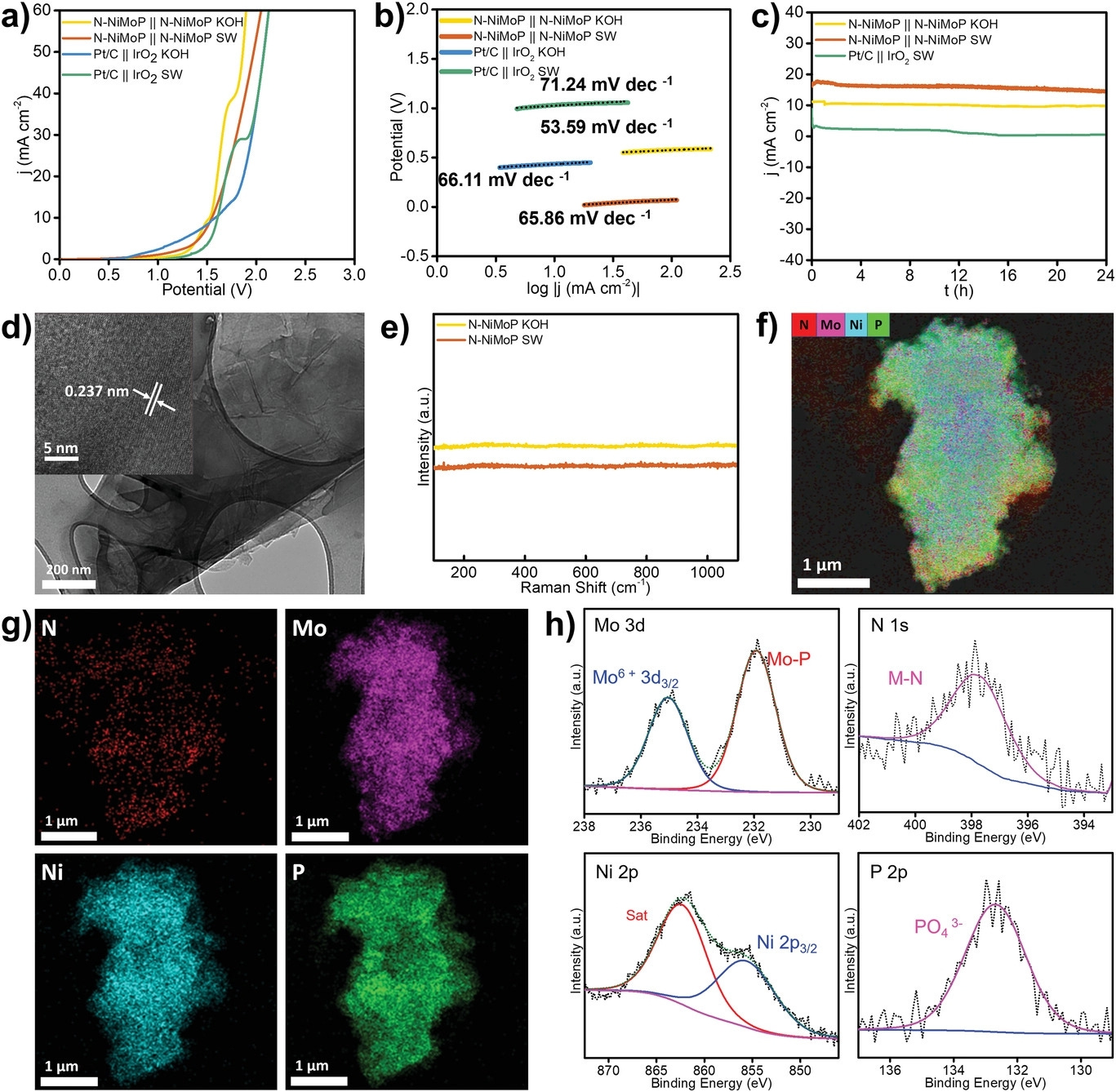
In the experiment, the researchers used the N-NiMo3P nanosheet catalyst to achieve complete water splitting in 1 molar potassium hydroxide and seawater, respectively, with a cell voltage of 1.52 and 1.55 volts, and reached a current density of 10 milliamps per square centimeter.
The researchers said that this technology is not only simple, scalable, but also more cost-effective than any “green” hydrogen production method on the market today, with an estimated cost of only 2 Australian dollars per kilogram of hydrogen.
What does this mean? Hydrogen has the highest energy density of all fossil fuels, 3 times that of gasoline, 3.9 times that of alcohol, and 4.5 times that of coke. The heat energy produced by burning one kilogram of hydrogen is 142.4 MJ, while oil, coal and natural gas are 44 MJ, 29 MJ and 50 MJ respectively.
The most most most critical thing is that the product of hydrogen combustion is only water, which does not produce any pollutants, and has no harm to the environment and human health. It can be said to be a complete crush on fossil fuels.
This means that if we can use seawater to produce hydrogen, we can get rid of our dependence on fossil energy, and achieve a low-carbon economy and cope with climate change.
This technology is expected to bring revolutionary changes to the green energy industry. Currently, the research team has applied for a technology patent and plans to develop an electrolysis cell prototype to produce hydrogen on a large scale.
You can find the principle and details of this technology in the paper in the appendix. If you are interested in seawater hydrogen production, you might want to read it and learn how this magic catalyst works.
Perhaps one day in the future, your car, airplane and household appliances might all be powered by seawater (hydrogen), and we can all enjoy the convenience of clean, low-carbon and high-efficiency energy, as well as the blue sky, white clouds and the spring breeze of nature.
Paper:
https://onlinelibrary.wiley.com/doi/10.1002/smll.202207310